Structural Formula - The Lewis Formula

Table of Contents
Elements combine to form chemical substances. Elements of the same type can bond with each other, but different elements can also form bonds. However, the formation of bonds always follows established laws.
Depiction
In reality, the resulting structures are arranged three-dimensionally, but for simplified representation, they are usually depicted as two-dimensional drawings. Several notations for representing structural formulas have been established. The simplest and probably most frequently depicted is the so-called Lewis formula. The term electron formula is also used as a synonym.
In the Lewis formula, each element is represented by its letter abbreviation. For example, O stands for oxygen and C for carbon. The same letters are used as in the periodic table.
Electrons are represented by a dot; if there are two electrons, as in a lone pair or a bond, this is represented by a line. It would also be permissible to draw two dots next to each other, but the line symbolizing two electrons has largely prevailed. If you ever see drawings with the dot form, it is usually in textbooks to illustrate the principle. To create a Lewis structural formula, you can use the octet rule as a guide.
Example
Let's take the example of water. Water has the molecular formula H2O. It consists of two hydrogen atoms bonding with one oxygen atom. Since hydrogen only has one outermost electron, also called the valence electron, it can only form one bond. Oxygen is in the sixth main group of the periodic table and therefore has six valence electrons. According to the octet rule, however, it would ideally like to have eight electrons in its outermost shell in order to achieve a stable and energetically favorable state. When the three atoms, i.e. two H and one O, combine to form water, they share their valence electrons to fulfill the octet rule. Note, however, that hydrogen only ever strives for two electrons. More cannot fit in the K shell. Its outermost shell is therefore already fully occupied.

To draw the structural formula for water, you need to consider how the atoms need to be arranged to fulfill the Octet Rule. Another tip is that hydrogen always appears on the outside, regardless of the compound, since it can only form one bond. So, draw an H for a hydrogen atom and a dot next to it for the hydrogen atom's electron. For oxygen to have eight valence electrons, it must share its electron with both hydrogen atoms, so it is drawn in the middle. Then you can add the six valence electrons as dots. However, you need to distribute these evenly. Therefore, always write them in a circle around the sides of the letter until you get the correct number. For oxygen, it would look like this:
A bond always consists of two electrons. So it's now easy to see where the two hydrogen atoms can bond. However, you don't draw the second electron in a plane next to the oxygen electron, but offset next to it, since the electron belongs to the hydrogen. Finally, you can connect all the electron pairs to form lines. You have now created the Lewis structure for water.
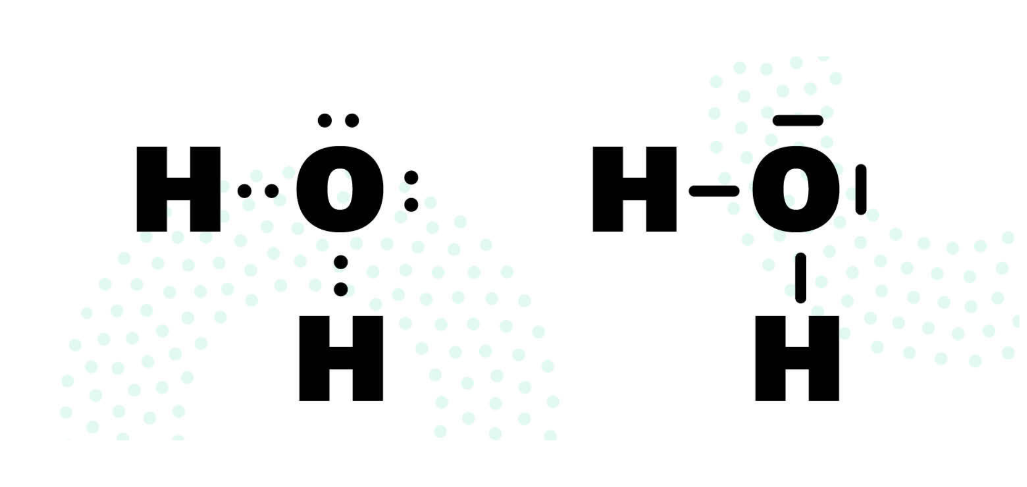
By following this methodical approach, you will arrive at the correct structural formula. Theoretically, a hydrogen atom could have been positioned to the right and left of the oxygen atom, and the Octet Rule would have been fulfilled. In reality, however, it looks like the diagram above. This is because these bond angles are energetically most favorable. So, even in compounds unfamiliar to you, you can see that you arrive at the correct, two-dimensional arrangement of atoms.
Also, note that electrons always want to be in pairs. Free electrons only exist in radicals; otherwise, electrons are always paired in a compound. If they are not involved in a bond with other atoms (see the above diagrams for the oxygen atom in water), they are called lone electron pairs.
Studydrive - Your companion for Studying
With Studydrive, you're always well prepared for your studies! Exchange ideas with other students, get peer-to-peer support for any questions you may have, don't miss anything shared between your classmates in the course, and find helpful study materials.