The best summaries

Chemistry - bonding.docx


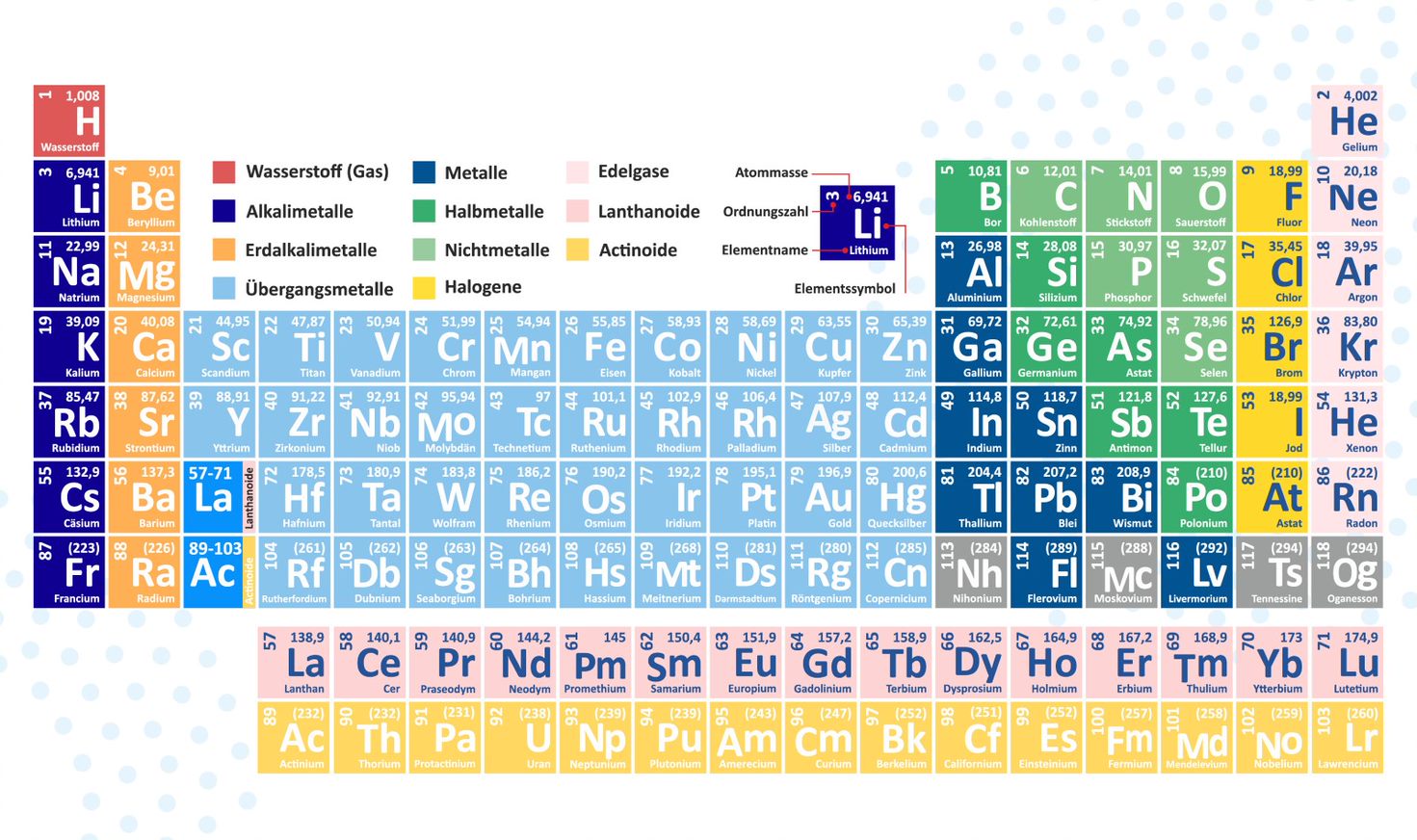
The periodic table is a chemist's most important tool in present day. Only with the help of this special table can certain trends and properties be identified. Likewise, many chemical reactions can be explained or even predicted with it.
The periodic table of elements serves as an organizational scheme and was first presented schematically by Dmitri Mendeleev and Lothar Meyer (independently of each other, but coincidentally at roughly the same time – 1869 – and very similarly). Over time, the periodic table made it possible to predict undiscovered elements because it follows a unique pattern. Likewise, some properties became clear and predictable for the first time, as they were repeated in a certain series of elements and could thus be applied to other elements.
You're familiar with the classic periodic table of elements from school. It's a table in which the elements are arranged according to simple rules. The chemical elements are arranged sequentially according to the number of protons in the atomic nucleus.
This number becomes the atomic number of the element. This number also indicates the number of electrons. This is because the elements are supposed to be neutral, and the positivity of the protons and the negativity of the electrons balance each other out.
As the atomic number increases from left to right, the elements are arranged one after the other in a "row”. The atomic number also reveals when the next row, the so-called period, begins. This is slightly more difficult to understand. Take a closer look at the atomic structure to understand when the next period begins.
It is important to note that certain properties repeat themselves with increasing atomic numbers. This is why the term "period" was chosen, and the whole thing was ultimately called the "periodic table."
When considering the structure of an atom, an atom is linked to a shell model. It is important to note that the atomic nucleus, consisting of protons and neutrons, is surrounded by shells. The surrounding shells can be schematically represented as circles around the nucleus.
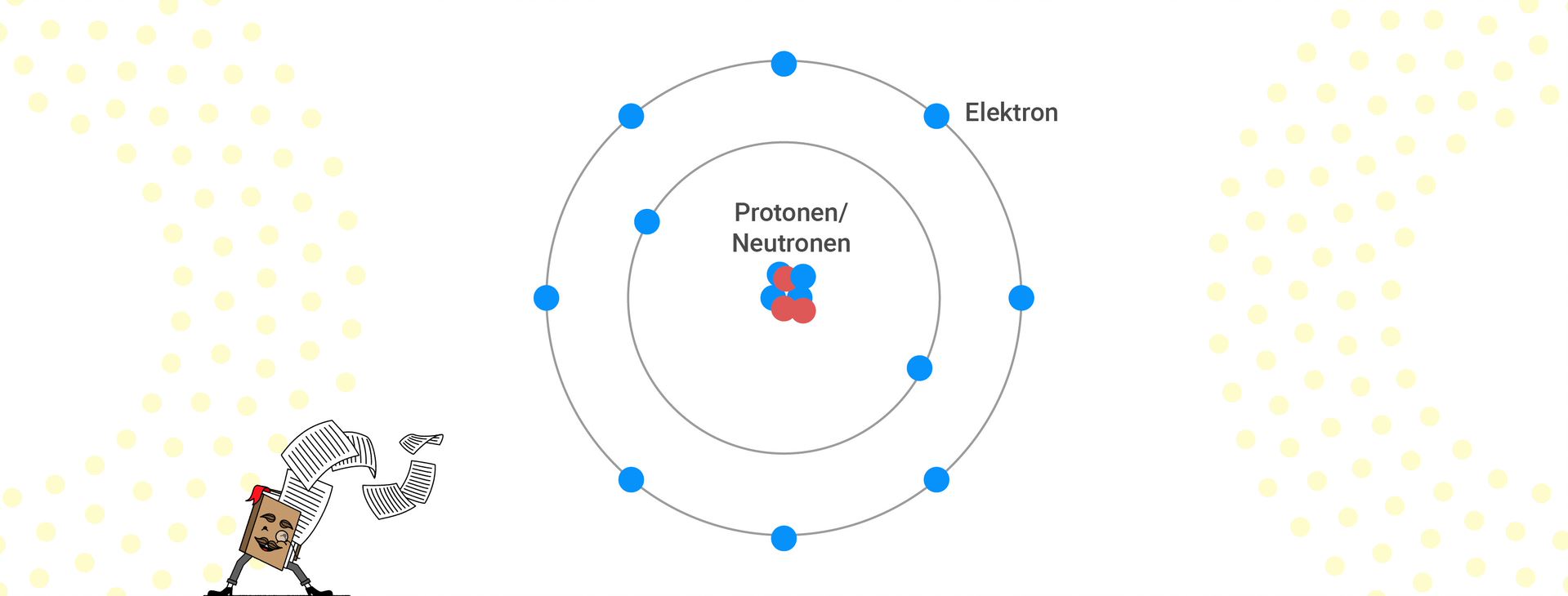
A maximum of two electrons are permissible in the first shell. The first shell represents the first period, in which hydrogen and helium are located. Eight electrons are permissible in the second shell, bringing us to the second period.
Eight electrons are allowed in the third and subsequent periods. Rule of thumb: The first shell has a maximum of two electrons; from the second shell onwards, a maximum of eight electrons are allowed. Each new shell forms a new period.
In addition to the periods, there are also groups. The groups represent the columns in the periodic table. It is clear that elements with chemically similar properties are grouped together. These groups are described and differentiated using various terms. A distinction can also be made between main groups and subgroups. Currently, we will only deal with the main groups.
The first group, excluding the hydrogen atom, is called the alkali metals. These are metals that form strong bases in reactions.
The second main group consists of alkaline earth metals. These, too, are alkaline metals, but like the earth metals, they are poorly soluble in water.
This brings us to the third main group, the earth metals or the Boron group. Boron metals are poorly soluble in water and are mistakenly called metals. Boron is not a metal, but rather a semimetal. The remaining elements are all metals.
The fourth main group comes next. It is named after the first element in it -- Carbon. It is commonly referred to as the Carbon group.
The fifth main group also bears the name of the first element in the group and is called the Nitrogen group. Nitrogen exists as a diatomic gas at room temperature.
The sixth main group, sometimes referred to as the oxygen group and more commonly known as chalcogens. Chalcogens are usually found in nature as minerals. The most well-known metal chalcogenides mostly consist of oxides and sulfides. Oxygen, as we know, is present in the air and is also a gaseous diatomic molecule. The remaining chalcogen elements are solid.
The seventh main group has a special name, and we often interact with it. The elements of the seventh main group are called halogens. These non-metals not only exhibit high reactivity in their elemental state, but also appear colored and form salts with metals. This is where the alkali and alkaline earth metals come into play again. Halogens also react with hydrogen to form hydrogen halides, which are monoprotic and classified as strong acids. Fluorine and chlorine form gaseous diatomic molecules. Bromine, on the other hand, is liquid. Iodine and astatine, however, are solid.
The last main group, and thus the eighth, contains the noble gases. In this category of gases, the last electron shells are either completely occupied by electrons or empty. This is also called the noble gas configuration because the atoms have full shells and thus do not undergo chemical reactions—or do so only under extreme conditions. Therefore, they do not exist as molecules (e.g., as diatomic gases), but rather as monatomic gases at room temperature.
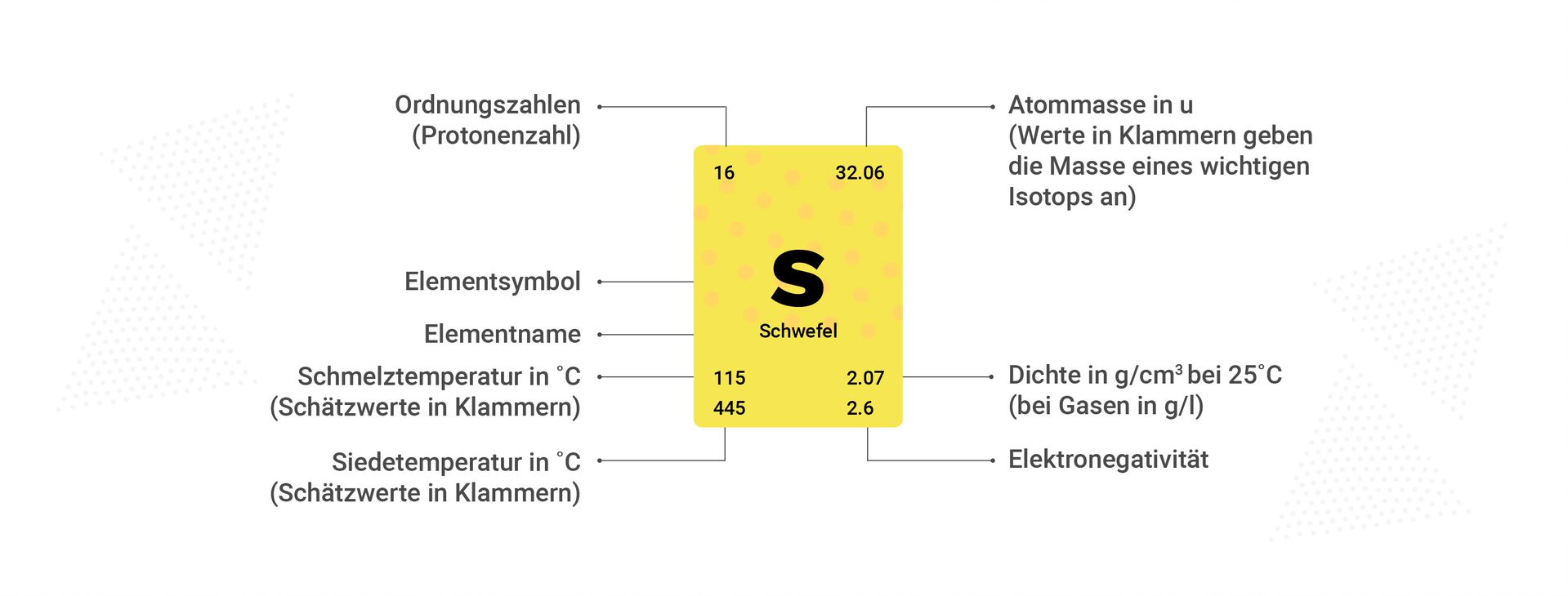
As explained in the beginning, the periodic table indicates the number of protons and electrons via the atomic number. The element symbol is an abbreviation for the element itself. The atomic mass of the element is usually also included in the grid, which can be calculated from the masses of protons, neutrons, and electrons. However, the mass of electrons is neglected when calculating the number of neutrons. One assumes 1 u for each proton or neutron.
For an atomic mass of approximately 12 u and 6 protons, the number of neutrons can be calculated by the difference, since the mass of protons is 1 u. Sometimes the electronegativity is also given, which is the measure of an atom's ability to attract electrons in a chemical bond. The bond (link) can usually also be predicted from the difference between the electron negativities of two different atoms. A distinction is made here between nonpolar, polar, and ionic.
In colored periodic tables, color coding also reveals properties such as metals, metalloids, and nonmetals. Furthermore, the periodic table indicates the number of shells. The number of main group elements can be used to predict the number of electrons in the outermost shell. These electrons have a special name because they participate in these reactions—they are called valence electrons.
On Studydrive, you'll find a wealth of learning materials to help you prepare for courses or exams. Here you'll find summaries and notes, solutions to past exams, and worksheets.



